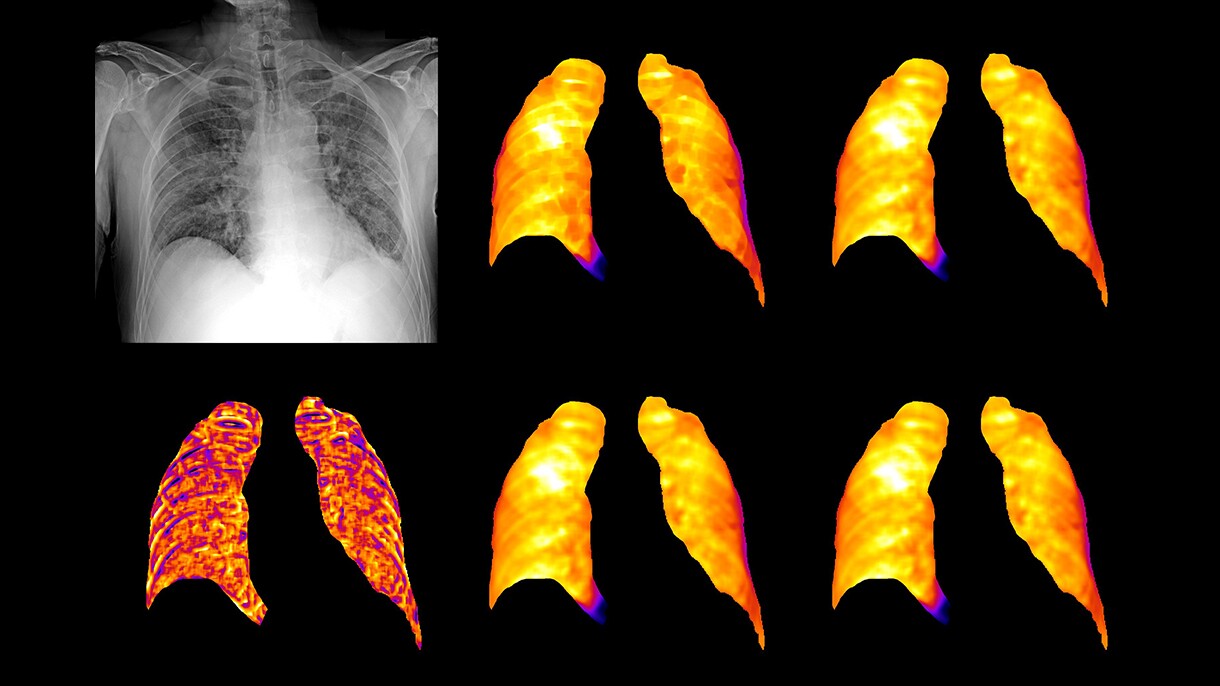2020 was a year like no other. By March, the COVID-19 virus had spread around the world, impacting families, businesses, and communities. Many are still fighting the spread of the virus, which has introduced several new variants that are threatening our communities. The speed at which the virus spread left COVID-19 diagnostics lagging and the healthcare community looking for new ways to use technology to help.
As countries grappled with the challenge of scaling COVID-19 testing, we launched the Amazon Web Services (AWS) Diagnostic Development Initiative to help organizations around the world apply the power of the cloud to accelerate diagnostics research and development. Through this initiative, AWS committed $20 million in computing credits and customized expertise from the AWS Professional Services team to support customers using AWS to drive diagnostic innovations.
In the first phase of the initiative, AWS helped 87 organizations in 17 countries ranging from nonprofits and research institutions to startups and large businesses. We have awarded $8 million supporting a range of diagnostic projects, including molecular tests for antibodies, antigens, and nucleic acids; diagnostic imaging; wearables; and data analytics tools that use artificial intelligence and machine learning to detect the virus.
01 / 03
As we launch the next phase, we are excited to broaden the AWS Diagnostic Development Initiative’s scope and distribute the remaining $12 million this year. Starting April 12, we are expanding the scope to three new areas: 1) early disease detection to identify outbreaks at the individual and at the community level; 2) prognosis to better understand disease trajectory; and 3) public health genomics to bolster viral genome sequencing worldwide.
AWS will prioritize COVID-19 projects but will also evaluate projects focused on other infectious diseases. We will accept applications through the end of the year, with priority consideration given to applications received before July 31. Interested organizations can apply here.
“We have seen transformative innovations in how we diagnose disease over the past year, from machine learning-powered X-ray imagery analysis to new developments in rapid, high quality, and direct-to-consumer tests,” said Dr. Vin Gupta, chief medical officer of Amazon’s COVID-19 Response. “These changes will continue to evolve and improve our ability to respond to future outbreaks. We have already seen inspirational results from the Diagnostic Development Initiative, and we look forward to supporting broader uses of cloud technologies to enable organizations and communities to identify and respond even faster to future outbreaks.”
As the pandemic worsened, we were able to build on a foundation of AWS technology that enabled us to help thousands of communities—millions of lives—all across the United States.
Jim Chou, vice president of engineering at Helix
Speeding customer innovation
The AWS Diagnostic Development Initiative has accelerated projects that are having an immediate impact on COVID-19 detection and changing what’s possible with medical diagnostics. These projects are not only helping the medical community rapidly respond to COVID-19, but also supporting important work for many other infectious diseases. Here are just a few of the projects funded by the AWS Diagnostic Development Initiative:
Stanford University School of Medicine develops smartwatch-based “alarm system” for diagnostics
Researchers at the Stanford University School of Medicine’s Healthcare Innovation Lab have developed a smartwatch app designed to correctly flag signs of a person’s immune system fighting a potential COVID-19 infection. The app is powered by an algorithm that detects changes in an individual’s resting heart rate and step count. Early results are promising, and a pilot trial successfully alerted newly infected individuals as early as 10 days before they became aware of any symptoms. The app has entered the next phase of study, and the Stanford team is recruiting participants with the goal of reaching 10 million participants to increase its ability to detect signs of COVID-19 in real time. This smartwatch-based early detection system was built on AWS with the support of the AWS Professional Services team, who collaborated with the researchers to help the study scale its data processing pipeline.
01 / 02
“We’re hopeful that ongoing screening using wearable devices can provide scalable diagnostics solutions to overcome current testing barriers, and that expanding data access to a broader range of researchers will contribute to new discoveries that improve human health," said Michael Snyder, Ph.D., professor and chair of genetics for Stanford University’s School of Medicine. "We look forward to continue pushing the boundaries of what is possible with the cloud.”
01 / 03
Illumina accelerates the identification of viral mutations through next-gen sequencing
Genomics is playing a fundamental role during the COVID-19 pandemic, and Illumina is driving the implementation of genomics through rapid innovation. The viral genome of SARS-CoV-2 was first sequenced on Illumina technology in China and made public on January 11, 2020. As part of a comprehensive strategy to bring rapid solutions to its customers and the global scientific community in the face of the pandemic, Illumina released its SARS-CoV-2 Data Toolkit in April 2020. The toolkit includes a number of apps built-for-purpose to analyze COVID-19 samples on its BaseSpace™ Sequence Hub, which runs on Amazon Elastic Compute Cloud (EC2). The focused analysis pipelines in the toolkit were made available to researchers at no cost for six months in 2020. As a result, Illumina has helped over 800 users process viral sequencing samples across its different apps. As genomic surveillance became more prominent toward the end of 2020, use of the toolkit’s DRAGEN COVID Lineage app—used to identify mutations in the viral genome—has escalated rapidly as health experts track for known and emerging variants that could inform public health decisions.
“Currently, many countries are reaching a point whereby public health agencies need to maintain an accurate catalog of the variants in their region, detect emerging strains, and assess any individual strain’s potential to impact vaccine effectiveness," said Phil Febbo, MD, chief medical officer of Illumina. "Our partners and customers rely on Illumina’s high-quality sequencing and data analytics to ensure accurate viral genomes and strain typing—which require immense computing power and processing. We value the innovative and scalable cloud-based infrastructure offered by AWS which enable critical worldwide cooperation—an essential factor in seeing the end to this pandemic.”
Helix ramps up high-sensitivity COVID-19 testing across the U.S.
Helix, a genomics startup that has built its entire software platform on AWS, is rapidly expanding access to high-sensitivity molecular COVID-19 testing with next-day turnaround time for health systems, employers, governments, and other organizations across the U.S. The Helix® COVID-19 Test has received Emergency Use Authorization from the U.S. Food and Drug Administration (FDA), and the company is scaling to process up to 100,000 COVID-19 tests per day in the U.S., making it one of the nation's largest COVID-19 testing laboratories. Helix provides an end-to-end, highly scalable solution to detect active COVID-19 infections by enabling easy self-collection of samples with lower nasal swabs, and a choice of unsupervised or supervised on-site sample collection.
“Working with AWS has enabled us to not only stand up a COVID-19 solution in short order, but also allowed Helix to scale quickly to meet a surge of testing demand this past winter. The impact of this is huge. As the pandemic worsened, we were able to build on a foundation of AWS technology that enabled us to help thousands of communities—millions of lives—all across the United States,” said Jim Chou, vice president of engineering at Helix.
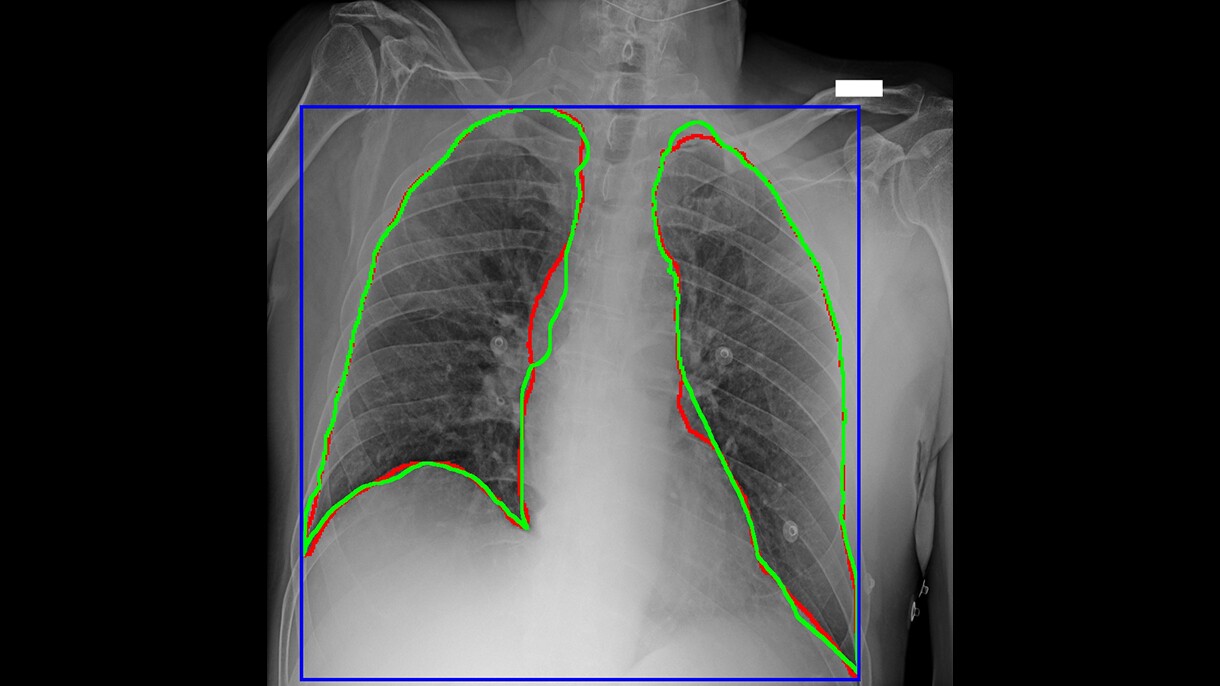
Centro Diagnostico Italiano and Bracco Imaging create open-source imaging archive project
A consortium of healthcare and research organizations led by Centro Diagnostico Italiano and Bracco Imaging collaborated to create the AIforCOVID Imaging Archive, a repository with almost 1,000 chest X-rays and anonymized clinical data of COVID-19 patients. Images and clinical data were analyzed using three machine learning approaches developed by the research organizations and performed on AWS. Based on these analyses, the consortium developed a model that is able to predict the clinical outcome of COVID-19 patients from chest X-ray images and clinical data. All data collected by the AIforCOVID network is available for download to members of the international scientific community from the project's website.
“Radiological imaging is playing a crucial role in COVID-19 patient management for diagnosis and treatment selection. Thanks to the power of machine learning delivered by AWS’s technology and professional support, we have been able to quickly make sense of the vast volume of data generated by diagnostic imaging and clinical analyses," said Sergio Papa, director of diagnostic imaging and stereotactic radiosurgery, Centro Diagnostico Italiano. "We are working to deliver a practical resource that hospitals and organizations around the world can apply to increase their understanding of the disease, facilitate their own diagnostic efforts, and improve patient treatment"
"Working with the Centro Diagnostico Italiano and AWS, we have begun exploring the potential of applying artificial intelligence to clinical data, and we realized that we are just starting to grasp the extent of this revolution," said Fabio Tedoldi, research and development director, Bracco Global Business Unit Imaging. "Healthcare is moving quickly toward new ways of integrating and interconnecting data with clinical practice and patient management."
Trending news and stories
- Amazon One Medical introduces weight management program with upfront GLP-1 medication costs
- How Amazon team members volunteer year-round to strengthen local communities
- A record number of independent sellers in Amazon’s store surpassed $1 million in sales in 2025
- Amazon's CEO of Worldwide Stores explains why telling your team to 'move faster' doesn't work